May 2022 | Volume 23 No. 2
Fighting Back against COVID-19
VACCINE BREAKTHROUGHS
Current vaccines for SARS-CoV-2 have done well protecting people from severe illness and death, but some studies have shown that even vaccinated people may continue to carry quite high levels of the virus in their nose, a situation that may underlie asymptomatic transmission and re-infection. In response, HKU researchers have developed new vaccines that can address that problem and are both cheap and easy to manufacture.
Professor Chen Honglin has drawn on his earlier work on a vaccine for influenza and coronaviruses to develop a nasal vaccine for COVID-19, while Professor Chen Zhiwei has adapted an HIV-AIDS vaccine he has been working on into an injectable COVID-19 vaccine. While work on their earlier vaccines is still going through the years-long process of pre-clinical and clinical trials, the urgency of COVID-19 has helped speed things up for the new vaccines and encouraged them to look at the effectiveness of combining the two.
After multiple tests, they found that the injectable vaccine followed by a booster from the nasal vaccine could offer protection against the virus and reduce the viral load in the nose in animal trials. The findings were published late last year in The Lancet’s online journal EBioMedicine.
“So far, this is the only vaccine combination able to block nasal infection and provide effective protection in both the upper and lower respiratory systems,” Professor Chen Honglin said. The nasal vaccine also has potential as a booster for people who have received Pfizer-BioNTech or other mRNA-based vaccines.
Building expertise
The origins of these vaccines date back two decades ago when, following the SARS outbreak in 2003, HKU began to build up expertise in infectious diseases. It is now a world leader in the field, hosting among other things the State Key Laboratory of Emerging Infectious Diseases, which is led by Professor Chen Honglin and includes team member Professor Chen Zhiwei, who also is Director of the AIDS Institute at HKU.
Before 2020, Professor Chen Honglin had been working on a vaccine candidate that showed potential for protection against both influenza and coronaviruses, by knocking out the NS1 gene from the NS segment in an influenza virus and inserting a receptor-binding-domain of the MERS (Middle East Respiratory Syndrome) coronavirus in its place. He quickly managed to replicate and insert SARS-CoV-2, which is also a coronavirus, and showed that it offered good protection against the virus.
He also decided to focus on developing his findings into a nasal vaccine that provides ‘frontline’ protection where the virus can enter the body. Although a nasal form takes longer to develop than other vaccines, it is cheap and easy to deploy and has the potential to be a second-wave vaccine to boost immunity. There is also growing interest in this form of vaccine, as exemplified in an article in The Washington Post in April this year headlined, “The next leap in coronavirus vaccine development could be a nasal spray”.
Professor Chen Zhiwei’s new vaccine is built on the model he developed for an HIV-AIDS vaccine. This DNA-based vaccine has been going through the conventional vaccine testing and approval process since 2013 and has a special way of delivering the antigen to antigen-presenting cells that induces potent antiviral immunity.
For SARS-CoV-2, he swapped the HIV gene for the COVID-19 virus and has found that on its own, this vaccine can provide protection similar to that of mRNA vaccines while being cheaper and easier to manufacture. It can also be easily adapted to new variants.

Professor Chen Honglin (left) has drawn on his earlier work on a vaccine for influenza and coronaviruses to develop a nasal vaccine for COVID-19.
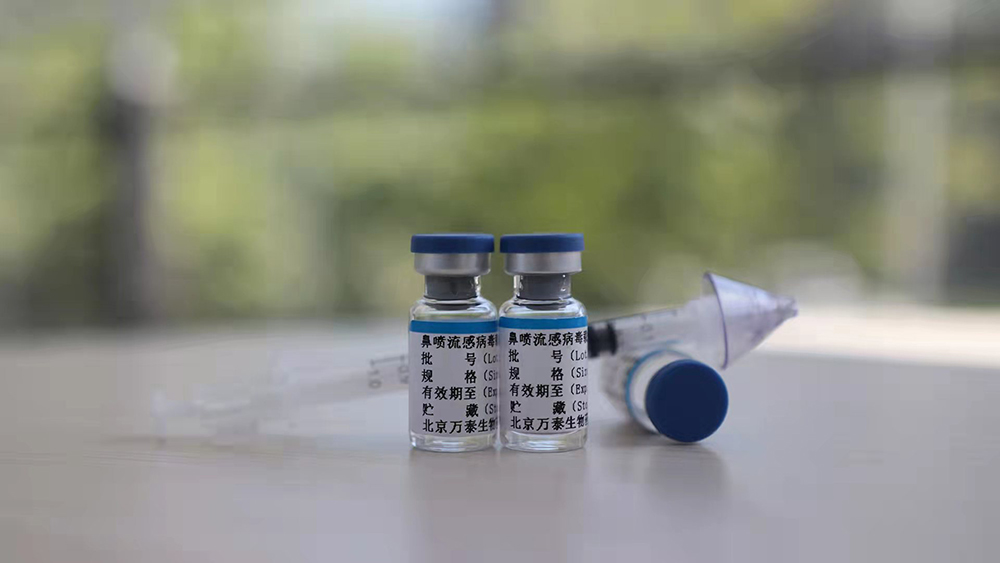
The nasal spray vaccine manufactured by Wantai BioPharm is currently in the third phase of trials in the Philippines, South Africa, Columbia and Vietnam.
So far, this is the only vaccine combination able to block nasal infection and provide effective protection in both the upper and lower respiratory systems.

PROFESSOR CHEN HONGLIN
Hurdles to development
The Lancet study reported on a variety of combinations using the two vaccines in animal models, with the best version being an injection followed later by the nasal vaccine. However, they still must complete safety tests individually before they can be tested together in humans.
The nasal vaccine is being developed with Wantai BioPharm, a Chinese pharmaceutical company, and is currently undergoing phase two and three clinical trials in the Philippines, South Africa, Columbia and Vietnam. The DNA-based vaccine has just finished a phase one trial that confirmed it is safe in humans. It is being developed in partnership with Immuno Cure Biotech Ltd in Hong Kong, which has worked with companies in the Mainland that have GMP [Good Manufacturing Practice] facilities for pre-clinical and clinical testing.
Both professors said the lack of GMP manufacturing in Hong Kong had slowed down their vaccines’ development because they had to look elsewhere for partners and testing facilities. Professor Chen Zhiwei hopes the government will reconsider its investment in this area, especially as other viruses are likely to threaten in future. When the first COVID-19 vaccines appeared, the government had to turn to sources outside the city in competition with other governments.
“We have the talent and expertise here with the State Key Laboratory and many scientists working on all kinds of viral diseases. Even when we can design a vaccine, the wait time from bench to human trial has been very long because of the disconnection in the middle and the lack of manufacturing capacity. I hope this situation can be changed so we can be prepared for future pandemics,” he said.

Professor Chen Zhiwei’s new injectable COVID-19 vaccine is built on the model he developed for an HIV-AIDS vaccine.
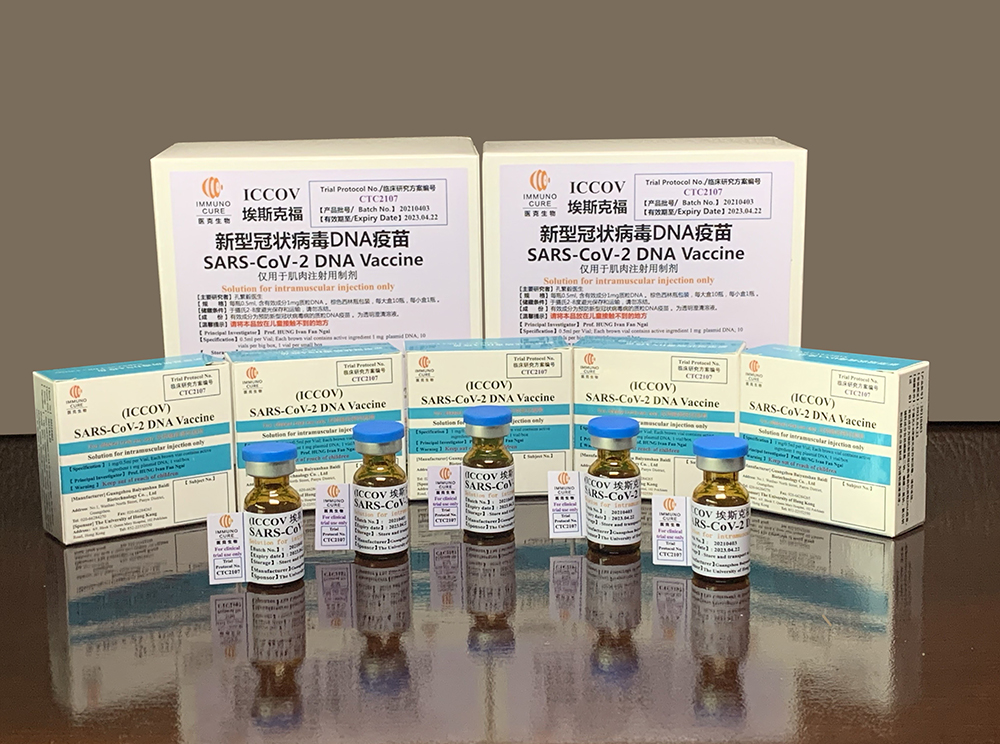
Developed in partnership with Immuno Cure Biotech Ltd in Hong Kong, the DNA-based vaccine has just finished a phase one trial that confirmed it is safe in humans.
The wait time from bench to human trial has been very long because of the disconnection in the middle and the lack of manufacturing capacity. I hope this situation can be changed.

PROFESSOR CHEN ZHIWEI
STEELING A MARCH
As COVID-19 continues to maintain its hold on Hong Kong, the development of a virus resistant stainless steel whose surface kills SARS-CoV-2 has big implications for use on frequently-touched areas such as lift buttons, door handles and handrails. E. coli and the H1N1 virus can also be inactivated on the surface of this special stainless steel.
The research, which was published in Chemical Engineering Journal, is a cross-disciplinary project led by Professor Mingxin Huang in the Faculty of Engineering’s Department of Mechanical Engineering, in collaboration with Professor Leo Lit Man Poon’s research team at the School of Public Health, HKUMed. Their work in this area began four years ago, before COVID had emerged.
“We started studying antibacterial and anti-H1N1 stainless steel and successfully invented antibacterial stainless steel containing silver (Ag) in 2019,” said Professor Huang. “When the COVID-19 pandemic began in early 2020, we changed our focus to anti-COVID-19 stainless steel. At the beginning, we thought we would be able to use our antibacterial steel for anti-COVID-19 purposes. But it failed, so then we invented our new anti-COVID-19 stainless steel that contains 20 per cent copper (Cu). The virus is killed by the Cu ions released from the steel surface.”

Sample of the anti-COVID-19 stainless steel that can kill the severe acute respiratory syndrome coronavirus 2 (SARS-Cov-2) on its surface.
Tuning stainless steel
The team achieved inactivation of pathogen microbes (especially the SARS-CoV-2) on the stainless steel surface by tuning the chemical composition and microstructure of regular stainless steel. The research also found interesting points about Ag and Cu as the alloying elements to prepare anti-pathogen stainless steel, namely that stainless steel containing Ag is good at killing bacteria, but cannot inactivate the virus, while stainless steel with a high Cu content (10 per cent or above) can effectively kill bacteria and virus.
Ordinary stainless steel has no inherent antimicrobial properties and studies have shown that the SARS-CoV-2 can remain stable and viable for as many as three days. Since crucial public areas such as hospitals and kitchens have myriad stainless steel surfaces, they also have a high possibility for virus transmission among people.
Said Professor Poon: “Recent data suggests that Omicron is more stable than the original SARS-CoV-2 on different environmental surfaces. This suggests that Omicron has an increased likelihood to be transmitted by fomite (i.e. objects or materials likely to carry infection, such as clothes, surfaces, utensils and furniture). The stainless steel developed by us can reduce such risk.”
Existing surfaces cannot be adapted to the new technology and would need to be replaced with the tuned stainless steel. Already there has been a lot of interest from the industry.
“Many local Hong Kong, Mainland, international companies have contacted HKU for potential use of our anti-COVID-19 steel,” said Professor Huang. “HKU Technology Transfer Office is talking to several companies regarding potential licensing and costs. If the negotiations go well, we may be able to see real products in the near future.
“The team has produced prototypes, including lift buttons made from the anti-COVID-19 stainless steel using mature powder metallurgy technologies. Its successful fabrication indicates a promising prospect for the production of future anti-COVID-19 stainless steel products.”
Existing technology
It is also viable in terms of production technology. “The stainless steel can be tuned using existing powder metallurgy technology, so it is quite ready for mass production,” Professor Huang continued.
Said Mr Litao Liu, the first author of the journal article and a PhD student supervised by Professor Huang: “Massive Cu-rich precipitates are permanently present in the stainless steel matrix. Thus, this anti- COVID-19 stainless steel can chronically inactivate pathogen microbes even though its surface is continuously damaged, meaning that even if the surface is scratched or dented, the inactivation of the virus microbes will continue.”
Summing up, Professor Huang said: “This anti-COVID-19 stainless steel can replace some of the frequently-touched stainless steel products in public areas to reduce the risk of accidental infection and help fight the COVID-19 pandemic. In addition, the findings on Ag and Cu as the allying elements to obtain antiviral and antibacterial properties have provided useful information for the future development of other forms of anti-pathogen stainless steel.”
The team has produced prototypes, including lift buttons made from the anti-COVID-19 stainless steel using mature powder metallurgy technologies. Its successful fabrication indicates a promising prospect for the production of future anti-COVID-19 stainless steel products.

PROFESSOR MINGXIN HUANG

